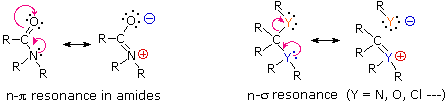The Pyranose Structure of Glucose
Establishing the Pyranose Structure of Glucose
The ring size of the cyclic hemiacetal structure assumed by many monosaccharides was determined by oxidative cleavage of a permethylated derivative. Five and six-membered rings are favored over other ring sizes because of their low angle and eclipsing strain. The equations in the following diagram illustrate this approach for the aldohexose, glucose.
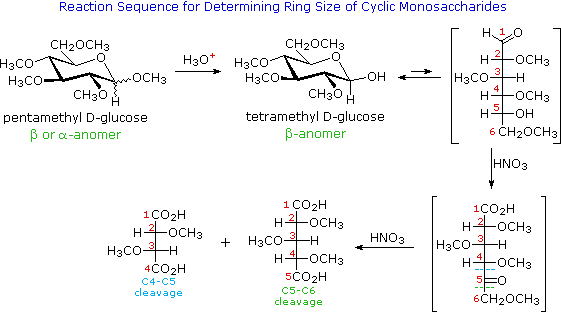
First, a pentamethyl derivative is prepared, as noted earlier. One of the methyl ether functions in this derivative is part of an acetal, and is therefore readily hydrolyzed by aqueous acid. The open chain form of this tetramethylglucose derivative is oxidized to a keto-acid intermediate by nitric acid treatment. The location of the ketone carbonyl reflects the size of the initial heterocyclic ring, C-4 for a pyranose ring and C-3 for a furanose ring. Further oxidation cleaves the carbon chain at bonds leading to the carbonyl group. This final oxidation produces a mixture of two dicarboxylic acids, differing in length by one carbon atom. Once these fragments have been identified, the location of the ketone function is established.
Chain Branching in Polysaccharides
Measuring Chain Branching in Polysaccharides
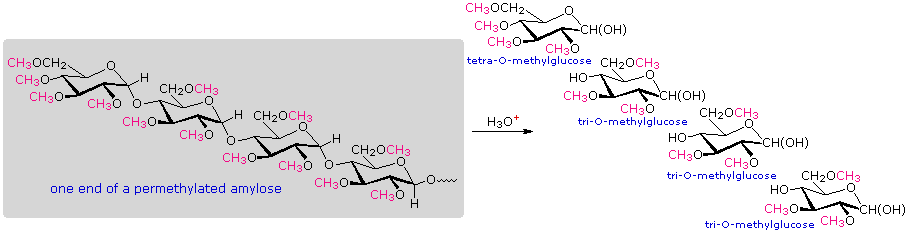
A simple strategy that provides easily interpreted results consists of methylating all the free hydroxyl groups in a polysaccharide, followed by hydrolysis of the glycoside bonds and determination of the methylated glucose products finally obtained. In the following diagram, the methyl groups added to an amylose chain are colored magenta. A chain of glucose units joined only by C-1 to C-4 glycoside bonds will give 2,3,6-tri-O-methylglucose as the chief product with a small amount of 2,3,4,6-tetra-O-methylglucose coming from the glucose unit on the chain's end. The shorter the average chain length, the greater is the relative amount of the tetramethyl derivative.
When chain branching of the kind found in amylopectin and glycogen is prevalent, 2,3-di-O-methylglucose is formed from each glucose at a branch point. The amount of this derivative formed is roughly the same as the tetramethyl derivative, and reflects the degree of chain branching. By clicking on the above diagram, a segment of permethylated amylopectin will be displayed and analyzed. The numbers presented in the diagram reflect that example. Typical results for amylopectin are 90% trimethylglucose + 5% dimethylglucose + 5% tetramethylglucose.
The Anomeric Effect
The Anomeric Effect
The equilibrium preference of most glucosides for the alpha-anomer is referred to as the anomeric effect. If we take away the extraneous functions and substituents from the glucoside structure, it is reduced to the 2-substituted pyran shown on the left in the following diagram. As the two conformational equilibria in the diagram demonstrate, the anomeric effect for a methoxy substituent is sufficient to change the preference for equatorial orientation (a consequence of steric hindrance) to a preference for the axial orientation. The anomeric effect in this case has been estimated at over 1 kcal/mol, but it should be noted that hydrogen bonding in hydroxylic solvents influences this factor.

One explanation for the anomeric effect focusses on dipole interactions
of the two ether functions. 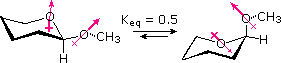 The dipole moments
associated with these divalent oxygens are similar to that of water. As
shown on the right, the ether dipoles in the equatorial conformer are
nearly parallel; whereas, in the axial conformer these dipoles are oriented
in opposite directions. Electrostatic interaction favors the latter
structure.
The dipole moments
associated with these divalent oxygens are similar to that of water. As
shown on the right, the ether dipoles in the equatorial conformer are
nearly parallel; whereas, in the axial conformer these dipoles are oriented
in opposite directions. Electrostatic interaction favors the latter
structure.
Another explanation centers on the interaction of nonbonding electron pairs
on oxygen with suitably oriented adjacent sigma bonds. Since this is a new
concept, a review of related neighboring electron interactions may be
helpful. We have noted that amides are the least reactive of the derivatives
of carboxylic acids, and this has been attributed to delocalization of
the nitrogen nonbonding electron pair by overlap with the pi-orbital of the
carbonyl group. This n-π interaction is illustrated by the resonance
equation on the left below. The right hand resonance equation shows an
equivalent n-σ interaction involving the general electron pair donor and
acceptor atoms designated Y and Y respectively.
To be precise, the nonbonding electron pair, n, is actually being
delocalized into the empty antibonding molecular
orbital associated with the π or σ-orbitals referred to above. As the
antibonding orbital becomes occupied (by electrons), the corresponding
bonding orbital is weakened and its electron pair moves onto the acceptor
atom. A vital factor in these interactions is the spatial requirement for
overlap between adjacent orbitals. For the amide example, this
stereoelectronic requirement will be illustrated by clicking on the above diagram. The lactam example on the
left is a typical amide. The basicity of the nitrogen electron pair is
dramatically
lowered (compared with an amine), and the carbonyl stretching frequency
in the infrared spectrum is likewise
lowered. Although the bicyclic compound on the right has a carbonyl
group adjacent to an amine, its properties are very different from the
amide on the left. The rigid bridged structure prevents the nonbonding
electron pair on nitrogen from overlapping with the pi- orbitals of the
carbonyl group. Indeed, if such an interaction occurred, the resulting
resonance contributor would violate Bredt's
rule. Not only is this keto-amine more basic than an amide, it is much
more reactive under base-catalyzed hydrolysis conditions than are most
amides, and the carbonyl stretching frequency is even higher than that of a
comparable ketone (thanks to the inductive effect of the nitrogen).
A similar stereoelectronic factor influences n-σ interactions, and is
presumably at the heart of the anomeric effect. The following diagram
serves to illustrate this feature for the 2-methoxypyran conformations.
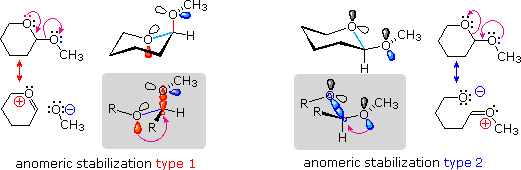
The resonance equations on the far left and far right show two kinds of
n-σ interactions, colored red for the endocyclic interaction on the left,
and blue for the exocyclic interaction on the right. When drawn as flat
Lewis structures the stereoelectronic characteristics of these interactions
are not evident, but in three-dimensional perspective drawings, such as
those near the center of the top row, they emerge. Again the red bonds and
orbitals define the endocyclic relationship, and the blue define the
exocyclic relationship. The partial structures drawn in the gray-shaded
boxes show the anti-periplanar orientation configuration required of the
interacting orbitals. The axial conformer on the left has two allowed n-σ
interactions (colored red and blue); whereas, the equatorial conformer on
the right has only one (colored blue). Furthermore, for pyran derivatives
of this kind the endocyclic (red) interaction is a permanent contributor to
the axial conformer, but the exocyclic (blue) interaction is only effective
for certain rotamer conformers of the methoxy group.
To examine a model of the axial
conformerClick Here.
The anomeric effect is also observed in acyclic systems, as the following comparison of butane and dimethoxymethane illustrates. The favored conformation changes from anti in the case of butane to gauche for the diether. The anomeric interaction in the latter compound is colored magenta.
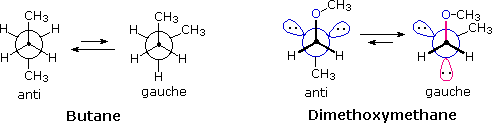
Donor and acceptor atoms other than oxygen may participate in the anomeric effect, with nitrogen being the best donor and halogen the best acceptor. Precise measurements confirm the changes in bond length expected for the anomeric effect. In the case of chloromethyl methyl ether (CH3-O-CH2-Cl), the CH2-O bond is 4% shorter than the CH3-O bond, and the C-Cl bond is 6% longer than the corresponding bond in ethyl chloride.
Modified Monosaccharides
Functionally and Structurally Modified Monosaccharides
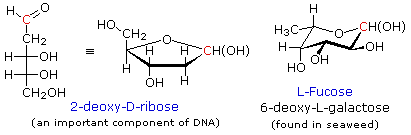
Many modified monosaccharides are deoxy-derivatives. In other words, one or more of the hydroxyl groups present in a normal sugar are missing. Examples of two such deoxy-sugars are given in the following diagram. In these and other formulas the anomeric carbon is colored red.
Amino sugars have an amino group, or several amino groups, replacing a customary hydroxyl group. Two examples will be displayed above by clicking on the diagram. D-Glucosamine, on the left, is probably the most common of the amino sugars. Its N-acetyl amide is the primary monosaccharide unit in the cellulose-like biopolymer that forms the exoskeletons of insects and shellfish. Finally, two examples of chain-lengthened sugars will be displayed by clicking a second time on the diagram. The structure of sialic acid, on the left, can be disconnected to reveal a glucosamine segment (colored blue) bonded to the methyl group of pyruvic acid. Further discussion of the role these saccharide derivatives play in biology is left to more specialized sources.
Vitamin C
Ascorbic acid, or vitamin C, is an important water soluble biological reducing agent that complements lipid soluble antioxidants such as vitamin E. Most animals synthesize this vital compound from glucose by a series of four enzymatic transformations, shown in the following diagram. Unfortunately, humans and a few other animals, including guinea pigs, some monkeys and many birds, do not have the enzyme, L-gulonolactone oxidase, needed for the fourth step. These animals must consume ascorbic acid as part of their daily diet or suffer chronic deficiency effects, which in extreme cases is the disease called scurvy. Although it does not have a free carboxyl group, ascorbic acid has a pKa of 4.17 as a consequence of vinylagous activation of the C-3 hydroxyl group (red hydrogen).
Sweetening Agents
Sweetening Agents
Sweetness is one of five types of taste sensed by humans. The others are
saltiness, sourness, bitterness and savouriness. It is generally regarded
as a pleasurable sensation, and the simple carbohydrates or sugars that
contribute to it are sought after and valued. Sucrose, from sugar cane and
sugar beets, is the standard sweetener used in western cuisine. A less
expensive alternative known as high fructose corn syrup (HFCS) was
developed in the late 1950's and is now widely used in baked goods and
beverages. HFCS is made from corn syrup by
enzymatic conversion of glucose to fructose. Since fructose is 2.3 times as
sweet as glucose and 75% sweeter than sucrose, HFCS provides a practical
substitute for sucrose in a variety of applications, and is available in
compositions ranging from 45 to 90% fructose.
Hydrolysis of sucrose produces an equimolar mixture of glucose and
fructose,that is sweeter than sucrose itself. Since the specific rotation
of these sugar solutions changes from +66.5º for pure sucrose to -22.0º for
the hydrolysis mixture (fructose is strongly levorotatory), the resulting
glucose fructose mixture is called invert sugar. It is widely used
in food manufacture in much the same way as HFCS. An enzyme, invertase,
which catalyzes the hydrolysis of sucrose in living organisms, is used in
the manufacture of invert sugar. Honey is similar to invert sugar,
consisting roughly of 38% fructose, 31% glucose, 9% disaccharides such as
maltose and 17% water.
Although there is a strong correlation between the rise of obesity in the
US and the use of HFCS for sweetening beverages and foods, it is not clear
whether this is a causal relationship. In fact, no substantial evidence
supports the idea that high-fructose corn syrup is responsible per se for
obesity. Instead, over-consumption of sugars, encouraged by the low cost of
HFCS and invert sugar, is the general culprit.
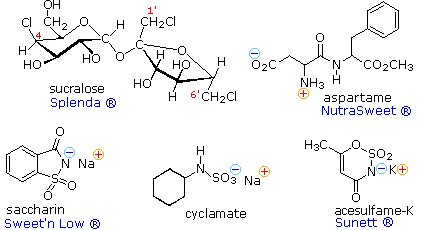
Because the synthetic sweeteners are many times sweeter than sucrose, only small amounts are needed to achieve a desired effect. Also, most are not significantly metabolized, so their use does not introduce additional calories into a diet. For some individuals, however, taste overtones such as bitterness reduce the suitability of these agents as sugar substitutes. If the sweetness of sucrose is taken as a standard, then these and other sweetening agents may be ranked accordingly, as listed below.
|
Compound |
sucralose |
saccharin |
acesulfame-K |
aspartame |
cyclamate |
fructose |
sucrose |
glucose |
maltose |
lactose |
|---|---|---|---|---|---|---|---|---|---|---|
|
Sweetness |
600 | 300 | 200 | 180 | 30 | 1.7 | 1.0 | 0.7 | 0.3 | 0.15 |
Aspartame is a dipeptide composed of two natural amino acids,
phenylalanine and aspartic acid, neither of which is sweet. Each of these
components has a stereogenic center, so four stereoisomers are possible.
The natural configuration of these amino acids is 2S, and (S,S)-Aspartame
is the commercial sweetener. The other three stereoisomers are not sweet,
and one is bitter. Aspartame undergoes a slow intramolecular acylation to a
cyclic dilactam that is not sweet. Consequently, soft drinks and other
beverages that contain dissolved aspertame have a limited shelf life. This
slow reaction is accelerated by heat, so aspertame is not suitable for
cooking purposes. To see an equation outlining the intramolecular acylation
click on the above diagram.
Acesulfame, cyclamate and saccharin are achiral, and are generally suitable
for cooking Since these compounds are acidic, their water soluble sodium or
potassium salts are the commonly used form. Sucralose has many stereogenic
centers, and although other chloro derivatives of sucrose are also sweet,
only the designated stereoisomer has been approved as a food
additive.
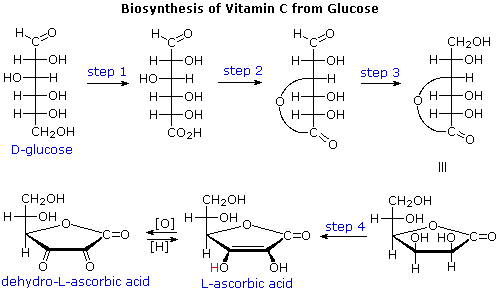
For sweetness to be perceived, molecules of a substance must activate
receptor sites in taste bud proteins on the tongue. This activation is
believed to occur when a molecule of suitable shape has a characteristic
functional distribution, referred to as the A, B, C system. According to
present theory, there are three essential components to a sweetener
molecule, oriented in a triangular fashion, as shown on the right. The A(H)
and B regions encompass functions of higher electronegativity, and the
distance between them must be greater than 2.4 A and less than 4.0 A. If
the distance between the atoms are not in this range then the substance
becomes bitter. The third part of this triangle, C, represents a
hydrophobic and lipophilic region of the molecule, not a specific atom or
group. This region does not bind to the receptor site. When a sweetener
molecule binds to a receptor, the AH region of the sweetener hydrogen bonds
to the B region of a receptor site, and the B region of a sweetener
hydrogen bonds to the AH region of the receptor site. This triggers a
response by cells in the taste bud, such that electrical impulses to the
brain create the perception of sweetness.
Acesulfame, saccharin and cyclamate are partially protonated by saliva,
generating the A(H) moiety. The adjacent SO2 group serves as the
B region in the first two cases. Similarly, the zwitterionic aspartic acid
segment of aspertame provides the A(H) and B sites for that sweetener.
Sucralose and natural sugars are not as easily analyzed, but more advanced
molecular orbital calculations have identified essential features in these
compounds.
To examine molecular models of these sweetening
agentsClick Here.
Safety
Before any food additives, including synthetic sweeteners, become
available for general use, they must be evaluated and approved by the FDA
(Food & Drug Administration). .First, the immediate or acute toxicity
of a substance is established by animal feeding studies. In this respect
all the sweeteners listed above were determined to be relatively innocuous
( roughly 10 times less toxic than salt and 100 times less toxic than
caffeine to rats ). It should be remembered that everything is poisonous
when taken in a sufficiently high dose. Thus, quantities of 25 to 30g of
caffeine in a single dose are lethal to most humans.
It is a relatively simple matter to establish the acute toxicity of a
substance, so most of the FDA's effort is directed to discovering chronic
toxicity and carcinogenic factors associated with a given agent. Such
studies are conducted in a variety of government, pharmaceutical and
academic laboratories, often over a relatively long time period, and
require care in the interpretation of results.
Saccharin, the oldest of the synthetic sweeteners, has been the subject
of more than 3000 separate studies. In the 1970's a report of increased
incidents of bladder cancer in rats fed very large amounts of saccharin led
the FDA to require that a warning label, "May be hazardous to your health."
be carried on products containing saccharin. Rat bladder cancer was also
reported in cyclamate feeding studies, and FDA approval was withdrawn in
1970. Later reviews suggested that the increased sodium load associated
with the very high doses in these studies may have been responsible for
some of the bladder cancer. Consequently, the warning requirement for
saccharin was lifted in 1991. Cyclamate is likely to be approved soon; over
600 studies have been conducted, and it is presently approved for use in 55
other countries.
Over 700 studies of aspartame are reported. Unlike saccharin, cyclamate and
acesulfame, which pass through our systems largely unmetabolized, aspartame
is converted to phenylalanine aspartic acid and methanol. The first of
these metabolites presents a danger to individuals having the genetic
disorder PKU, which inhibits their ability to further metabolize this
essential amino acid. Some concern has also been voiced concerning the
methanol metabolite, but the small amounts produced in normal use are
unlikely to pose a serious health issue (methanol is only 10% of
aspartame). Aspartame received FDA approval in 1981. It is worth noting
that there seem to be more anecdotal reports of health problems associated
with aspartame than with the other sweetening agents, but this may only
reflect its widespread use in soft drinks.
Sucralose, the newest and least studied sweetener (ca. 70 reports),
received FDA approval in 1998. Although largely unmetabolized, over 25% of
ingested sucralose is believed to be retained or metabolized to
dichlorofructose. Reports of thymus gland shrinkage and liver enlargement
will need to be investigated before being dismissed.
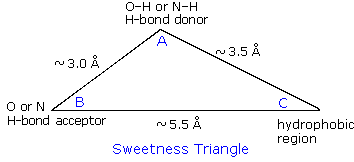
Stevia Glycosides
Stevia is a genus of herbs and shrubs native to subtropical and tropical
South America and Central America. The leaves of the plant Stevia
rebaudiana Bertoni have a sweet taste resulting from glycosides of the
diterpene steviol (structure on the right). 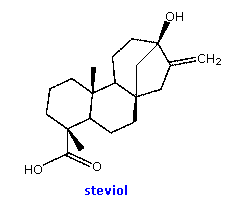 Stevioside and rebaudioside
A, the primary components, are glucosides attached to the hydroxyl
functions of steviol. They are heat stable, pH stable, and do not ferment.
Stevioside has a sweetness 200 to 350 times that of sucrose, and a relative
caloric value 300 fold less. Since stevioside does not induce a glycemic
response when ingested, it offers potential as a natural sweetener for
diabetics and others on carbohydrate-controlled diets.
Stevioside and rebaudioside
A, the primary components, are glucosides attached to the hydroxyl
functions of steviol. They are heat stable, pH stable, and do not ferment.
Stevioside has a sweetness 200 to 350 times that of sucrose, and a relative
caloric value 300 fold less. Since stevioside does not induce a glycemic
response when ingested, it offers potential as a natural sweetener for
diabetics and others on carbohydrate-controlled diets.
In South America, stevia leaves have been employed in ethnomedical
applications for centuries. In Japan, stevia extracts have been used as a
sweetener for over thirty years with no reported harmful effects.
Nevertheless, in 1991, at the request of an anonymous complaint, the United
States Food and Drug Administration (FDA) labeled stevia as an "unsafe food
additive" and restricted its import. The FDA's stated reason was
"toxicological information on stevia is inadequate to demonstrate its
safety." The 1994 Dietary Supplement Health and Education Act forced the
FDA in 1995 to revise its stance to permit stevia to be used as a dietary
supplement, although not as a food additive – a position that seems
contradictory because it simultaneously labels stevia as safe and unsafe,
depending on how it is sold.
Additional studies have shown that stevia improves insulin sensitivity in
rats, possibly promoting insulin production. Also, preliminary human
studies suggest that stevia may help reduce hypertension. Despite other
research pointing to the safety of stevia, government agencies continue to
express concern over a lack of conclusive evidence on this subject.
Oligosaccharides
Tri and Higher Oligosaccharides
Complex oligosaccharides are common components of numerous biologically
important macromolecules. In many of these systems aminosaccharides,
deoxysaccharides and C9 glyconic acids are found linked to more
common sugar units, so an amazing diversity of similar but distinct
structures exists. In this discussion we shall limit our attention to
relatively simple molecules composed of simple aldohexose units.
Appreciable amounts of oligosaccharides are found in certain foods, such as
peas and beans. The structural formulas of three such compounds are given
in the following diagram. They are all non-reducing sugars. A common
sucrose moiety is seen for the two rings on the right, and this is joined
to one or more galactopyranose rings by alpha-glycoside bonds at C-6. Two
enzymes are required to hydrolyze these oligosaccharides into
monosaccharides that are easily absorbed into the blood stream. The
galactose units are cleaved by alpha-galactosidase, and the
glucose-fructose link in sucrose is hydrolyzed by sucrase (or
invertase). Humans do not have a source of alpha-galactosidase in their
digestive system, so the oligosaccharide passes largely unchanged into the
colon. Anaerobic microorganisms in the colon ferment these sugars,
producing carbon dioxide and methane, gases that cause flatulence.
Cyclodextrins
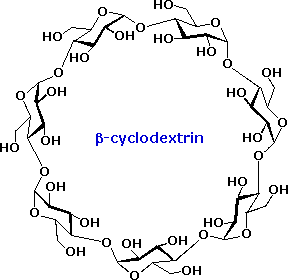
An interesting class of non-reducing oligosaccharides composed of glucopyranose rings joined 1-4 by alpha-glycosidic bonds are called cyclodextrins. Cyclodextrins are formed when starch is treated with an amylase enzyme from Bacillus macerans. Depending on the number of glucose units in the ring the cyclodextrins are named alpha (6), beta (7), and gamma (8). The shape of the cyclodextrins is that of a tapered ring, with the C-2 and C-3 hydroxyl functions on one edge and the CH2OH groups hanging from the opposite edge. The structure of the beta-isomer is shown on the right. By clicking on this structure a model of this cyclodextrin will be displayed. Because the interior of the cyclodextrin ring is relatively hydrophobic, these remarkable compounds are able to encapsulate small nonpolar molecules. They have been used as catalysts and aqueous transport agents.