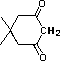|
This page has acidity tables for heteroatom organic acids and carbon acids. |
|---|
Because of the very large range of acid strengths ( greater than 1040 ), a logarithmic scale of acidity ( pKa ) is normally employed. Stronger acids have smaller or more negative pKa values than do weaker acids. A discussion of acid-base terminology is available here.The pKa values given here are extrapolated for water at 25 ºC. Many of the pKa values given for weak carbon acids are estimated from the rates of hydrogen exchange with very strong bases.
Other collections of pKa values include those compiled by Prof. D.A. Evans (Harvard); a large list compiled by W.P Jencks & F.H. Westheimer, presented by R. Williams (Penn. State); large collections of aqueous pKa values from ZirChrom and Kaye&Laby; and a collection of pKa measurements in DMSO solution, compiled by Prof. F.G.Bordwell and presented by Prof. H.J.Reich (Wisconsin).
Ionization Constants of Heteroatom Organic Acids
|
Common Name |
Formula |
Acidity Constant |
pKa |
|
trifluoromethanesulfonic acid |
CF3SO3H |
ca. 1013 |
ca. -13 |
|
benzenesulfonic acid |
C6H5SO3H |
ca. 103 |
ca. -2.5 |
|
methanesulfonic acid |
CH3SO3H |
ca. 3 * 102 |
ca. -2.0 |
|
trifluoroacetic acid |
CF3CO2H |
1.0 |
0.0 |
|
picric acid |
(O2N)3C6H2OH |
0.5 |
0.3 |
|
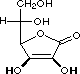
squaric acid |
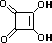 |
K1 = 0.33 |
1.5 |
|
trichloroacetic acid |
CCl3CO2H |
0.23 |
0.77 |
|
oxalic acid |
(CO2H)2 |
K1 = 6.5 * 10-2 |
1.2 |
|
dichloroacetic acid |
CHCl2CO2H |
5.5 * 10-2 |
1.25 |
|
fluoroacetic acid |
FCH2CO2H |
2.5 * 10-3 |
2.6 |
|
chloroacetic acid |
ClCH2CO2H |
1.36 * 10-3 |
2.87 |
|
citric acid |
C(OH)(CH2CO2H)2CO2H |
K1 = 7.4 * 10-4 |
3.13 |
|
formic acid |
HCO2H |
1.77 * 10-4 |
3.75 |
|
ascorbic acid |
|
K1 = 6.7 * 10-5 |
4.17 |
|
benzoic acid |
C6H5CO2H |
6.3 * 10-5 |
4.20 |
|
acetic acid |
CH3CO2H |
1.77 * 10-5 |
4.75 |
|
thiophenol |
C6H5SH |
2.5 * 10-7 |
6.6 |
|
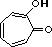
tropolone |
|
2.0 * 10-7 |
6.7 |
|
p-nitrophenol |
O2NC6H4OH |
5.7 * 10-8 |
7.2 |
|
peracetic acid |
CH3COO2H |
5.7 * 10-9 |
8.2 |
|
succinimide |
(CH2CO)2NH |
2.5 * 10-10 |
9.6 |
|
phenol |
C6H5OH |
10-10 |
10.0 |
|
chloral hydrate |
CCl3CH(OH)2 |
10-10 |
10.0 |
|
benzenesulfonamide |
C6H5SO2NH2 |
8 * 10-11 |
10.1 |
|
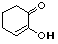
2-hydroxy-2-cyclohexenone |
|
5.0 * 10-11 |
10.3 |
|
ethanethiol |
C2H5SH |
2.5 * 10-11 |
10.6 |
|
acetoxime |
(CH3)2C=NOH |
6 * 10-13 |
12.2 |
|
2,2,2-trifluoroethanol |
CF3CH2OH |
4 * 10-13 |
12.4 |
|
imidazole |
C3H3N2H |
3.3 * 10-15 |
14.5 |
|
pyrrole |
C4H4NH |
10-15 |
15 |
|
ethanol |
C2H5OH |
10-16 |
16 |
|
1°-amides |
RCONH2 |
10-17 |
17 |
|
p-nitroaniline |
O2NC6H4NH2 |
3.3 * 10-19 |
18.5 |
|
t-butanol |
(CH3)3COH |
10-19 |
19 |
|
aniline |
C6H5NH2 |
10-27 |
27 |
|
1,1,1,3,3,3,-hexamethyldisilazane |
[(CH3)3Si]2NH |
10-30 |
30 |
|
pyrrolidine |
C4H8NH |
10-32 |
32 |
|
diisopropylamine |
[(CH3)2CH]2NH |
1.9 * 10-36 |
35.7 |
|
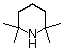
tetramethylpiperidine |
|
10-37 |
37 |
|
Michigan State University |
Ionization Constants of Carbon Acids
|
Common Name |
Formula |
Acidity Constant |
pKa |
|
tricyanomethane |
CH(CN)3 |
105 |
-5 |
|
dinitromethane |
CH2(NO2)2 |
2.5 * 10-4 |
3.6 |
|
5,5-dimethyl-1,3-cyclohexadione |
|
1.6 * 10-5 |
4.8 |
|
malonaldehyde |
CH2(CHO)2 |
10-5 |
5.0 |
|
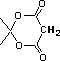
Meldrum's acid |
|
8 * 10-6 |
5.1 |
|
2,4-pentanedione |
CH2(COCH3)2 |
1.26 * 10-9 |
8.9 |
|
hydrocyanic acid |
HCN |
6.3 * 10-10 |
9.2 |
|
nitromethane |
CH3NO2 |
6.3 * 10-11 |
10.2 |
|
malononitrile |
CH2(CN)2 |
10-11 |
11.0 |
|
ethyl acetoacetate |
CH3COCH2CO2C2H5 |
10-11 |
11.0 |
|
tris(trifluoromethyl)methane |
(CF3)3CH |
10-11 |
11.0 |
|
bis(methylsulfonyl)methane |
CH2(SO2CH3)2 |
2.0 * 10-13 |
12.7 |
|
diethyl malonate |
CH2(CO2C2H5)2 |
3.2 * 10-14 |
13.5 |
|
bromoform |
Br3CH |
3 * 10-14 |
13.7 |
|
chloroform |
Cl3CH |
3.2 * 10-16 |
15,5 |
|
cyclopentadiene |
10-16 |
16.0 |
|
|
aldehydes |
RCH2CHO |
10-17 |
17 |
|
cyclohexanone |
10-17 |
17 |
|
|
phenylacetylene |
C6H5C≡CH |
10-19 |
19.0 |
|
indene |
|
10-20 |
20 |
|
methyl ketones |
RCOCH3 |
10-20 |
20 |
|
tris(phenylthio)methane |
(C6H5S)3CH |
10-23 |
23 |
|
methylsulfones |
RSO2CH3 |
10-25 |
25 |
|
alkyl esters |
RCH2CO2C2H5 |
10-25 |
25 |
|
nitriles |
RCH2CN |
10-25 |
25 |
|
acetylene |
HC≡CH |
10-25 |
25 |
|
fluroform |
F3CH |
10-28 |
28 |
|
methylsulfoxides |
RSOCH3 |
10-30 |
30 |
|
1,3-dithiane |
(CH2)3S2CH2 |
10-31 |
31 |
|
triphenylmethane |
(C6H5)3CH |
3.2 * 10-32 |
31.5 |
|
diphenylmethane |
(C6H5)2CH2 |
3.2 * 10-34 |
33.5 |
|
1-methylnaphthylene |
1-C10H7CH3 |
10-37 |
37 |
|
2-methylnaphthylene |
2-C10H7CH3 |
3.2 * 10-38 |
37.5 |
|
toluene |
C6H5CH3 |
10-40 |
40 |
|
benzene |
C6H6 |
10-43 |
43 |
|
ethene |
C2H4 |
10-44 |
44 |
|
cyclopropane |
C3H6 |
10-46 |
46 |
|
ethane |
C2H6 |
10-48 |
48 |
|
Michigan State University |