The Nature of Energy
The term energy is commonly used in many contexts, but seldom in a carefully defined way. A speeding automobile has considerable kinetic energy, which is transformed into heat energy and work (the rupture of metal and plastic components) if the automobile crashes into a stone wall. The accumulated snow on a steep mountain has considerable potential energy which is transformed in an avalanche, first into kinetic energy, and then into heat and work as the avalanche engulfs forests and towns and eventually comes to a stop. In these examples the potential and kinetic energy are directed (oriented) in space, whereas the heat produced consists of random molecular motion.
Chemists are concerned with both physical and chemical change, and the
energy associated with each. Chemical reactions involve the making and
breaking of covalent bonds, and these transformations have energy
consequences. Heat is the most common form of energy introduced into
or released from chemical reactions, but light, electrical current and
sound may also be involved. Also, if gases are produced in a reaction, the
work done on the surroundings by volume expansion becomes part of the
energy balance, and in explosive reactions may be the major consequence. Of
course, reactions that consume a gas undergo an opposite volume change, and
work is done on the system by the surroundings.
The traditional unit of heat energy used by organic chemists is the
calorie, defined as the heat needed to raise the temperature of 1
gram of water 1º C (from 14.5 to 15.5º C). Note that this calorie is not
the same as the Calorie used for nutritional purposes. In 1960 a revised
metric system based on seven fundamental units was established, and the
resulting SI unit of energy is the joule (J). The joule is a
unit derived from three fundamental units (mass, length and time), and is
used to express both heat and work. Both the joule and the calorie are
small, so it is common to express heats of reaction in kilocalories (kcal)
or kilojoules (kJ) per mole. To interconvert calories (or kcal) and joules
(or kJ) the following factors may be used:
|
1 cal = 4.184 J |
1 J = 0.2388 cal |
|---|
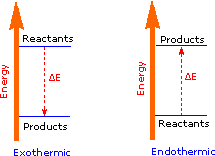
The covalent bond of a chlorine molecule provides a simple example of the energy changes associated with bond breaking and bond making. This bond may be broken by the introduction of heat or light energy, and it has been determined that 57.9 kcal/mol (242.3 kJ/mol) is required for bond homolysis. This is shown in the first of the following reactions; the second reaction describes the reverse bond-forming process. The energy absorbed or released in these reactions is referred to as the bond dissociation energy. If the bond dissociation energy is introduced from the surroundings in the form of heat, the transformation is said to be endothermic. If heat passes from the system to the surroundings, the transformation is termed exothermic. Using our initial terminology, we may say that the covalently bonded system has a lower potential energy than the unbonded diatomic system. Indeed, It is helpful to think of exothermic reactions as proceeding from a higher energy (less stable) reactant state to a lower energy (more stable) product state, as shown in the diagram on the right. Some basic principles of reaction energetics were discussed earlier.
|
Cl_Cl + heat____> 2Cl • |
An Endothermic Transformation |
 |
|---|---|---|
|
2Cl • ____> Cl_Cl + heat |
An Exothermic Transformation |
In more complex chemical reactions some (or even all) of the bonds that hold together the atoms of reactant and product molecules may be broken while other bonds are formed. Energy is required to break bonds, and since different bonds have different bond dissociation energies, there is often a significant overall energy change in the course of a reaction. In the combustion of methane, for example, all six bonds in the reactant molecules are broken, and six new bonds are formed in the product molecules (equation 1). To analyze such reactions we need to keep track of and evaluate heat changes in a precise and systematic manner.
|
(1) CH4 + 2 O2 |
 |
CO2 + 2 H2O + heat |
|
Reactants |
Products |
Thermodynamics
1. Enthalpy
The science that investigates the passage of energy from one system to
another, and the transformation of energy from one form to another is
called thermodynamics. Classical thermodynamics is a statistical
science in which observations are made on macroscopic samples. Heat energy
or heat content is designated as enthalpy, symbol H. The
enthalpy change in a physical or chemical transformation is written
ΔH, defined as Hproducts - Hreactants.
Because of this formalism, which holds for other thermodynamic functions as
well, exothermic reactions have a negative ΔH, and
endothermic reactions have a positive ΔH. Careful calorimetric
measurements of the heat produced by the combustion of methane (equation 1
above) establish a ΔH = -212.8 kcal/mol for this reaction.
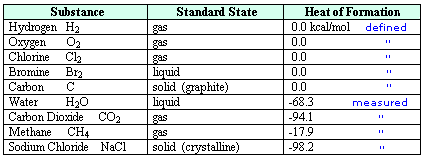
In order to consolidate and make use of thermochemical data of this kind, a
standard state has been defined. This is taken to be the form in
which a substance is stable at 25º C and one atmosphere pressure (760
torr). For solutions the standard state is a 1M concentration. The standard
states of some typical substances are listed in the following table.
Care must be taken to identify the most stable phase or form a given
substance will assume under standard conditions. In the case of water this
is the liquid phase, and for carbon it is graphite, not diamond. The
relative enthalpy of different compounds may then be given as a heat of
formation, symbol ΔHºf, as noted in the right hand
column. By definition, the heat of formation of an element in its
standard state is 0.0 kcal/mol. Reactions involving elements are then
used to determine the ΔHºf of the compounds produced. A small
degree sign to the upper right of the enthalpy symbol (e.g. Hº)
designates a measurement made under standard state conditions.
For examples of direct and indirect determinations of heats of formation
click on the above table. The first two
equations show how the heats of formation of water and carbon dioxide are
measured. A second click on the diagram displays an indirect method for
determining the ΔHºf of methane.
2. Bond Energies
Most heats of formation are negative, reflecting the strong covalent
bonds and lower enthalpy that characterizes stable compounds relative to
their elements. However, some stable compounds are found to have positive
heats of formation, e.g. benzene and condensed ring aromatics, making it
unwise to generalize from this data alone. As we have noted, heats of
reaction reflect the bond dissociation energies of bonds that are broken
and formed in the reaction, but the formalism of setting elemental heats of
formation to zero obscures the covalent bond dissociation energies of
diatomic elements such as H2, O2, N2 and
Cl2. Elements that have solid standard states (e.g. carbon)
present an even more complex bond dissociation energy challenge.
Fortunately, it is possible to determine the bond dissociation energy of
diatomic elements and compounds with precision by non-thermodynamic
methods, and together with thermodynamic data such information permits a
table of average bond
energies to be assembled. These bond energies or bond dissociation
enthalpies are always positive, since they represent the endothermic
homolysis of a covalent bond. It must be emphasized that for the common
covalent bonds found in polyatomic molecules (e.g. C-H and C-C) these are
average dissociation enthalpies, in contrast to specific bond
dissociation enthalpies determined for individual bonds in designated
compounds. Factors such as hybridization, strain and conjugation may raise
or lower these numbers substantially.
Common sense suggests that molecules held together by strong covalent bonds
will be more stable than molecules constructed from weaker bonds.
Previously we defined bond dissociation energy as the energy required to
break a bond into neutral fragments (radicals or atoms). The sum of all the
bond energies of a molecule can therefore be considered its atomization
energy, i.e. the energy required to break the molecule completely into
its component atoms. If this concept is applied to the reactants and
products of a reaction, it should be clear that a common atomization state
exists, and that the total bond energies of the reactants compared with the
bond energies of the products determines the enthalpy change of the
reaction. Thus, if the products have a greater total bond energy than the
reactants the reaction will be exothermic, and the opposite is true for an
endothermic reaction. The following diagram illustrates this relationship
for the combustion of methane. Always remember, a bond energy is energy
that must be introduced to break a bond, and is not a component of a
molecule's potential energy.
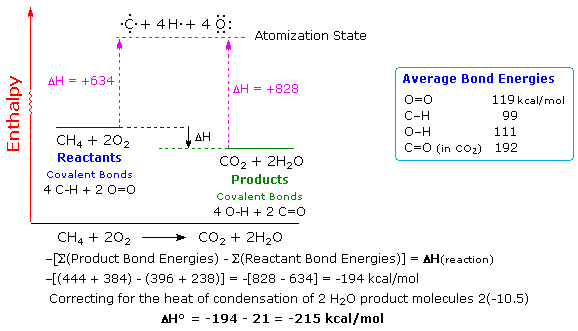
Bond energies may be used for rough calculations of enthalpies of
reaction. To do so the total bond energies of the reactant molecules must
be subtracted from the total bond energies of the product molecules, and
the resulting sign must be changed. This operation is outlined above for
the combustion of methane. To compare such a calculation with an
experimental standard enthalpy of reaction, correction factors for heats of
condensation (or fusion) must be added to achieve standard state
conditions. In the above example, gaseous water must be condensed to the
liquid state, releasing 10.5 kcal/mol of heat. Once this is done, a
reasonably good estimate of the standard enthalpy change is
obtained.
It may be helpful to note that the potential energy of a given molecular
system is inversely proportional to its total bond energies. In this sense
the potential energy of the methane + oxygen reactants is greater than that
of the carbon dioxide + water products. In this reaction, potential energy
is lost by conversion to kinetic (heat) energy.
3. Activation Energy
Thermodynamic calculations and arguments focus only on the initial and
final states of a system. The path by which a change takes place is not
considered. Intuitively, one might expect strongly exothermic reactions to
occur spontaneously, but this is usually not true. For example, the methane
combustion described above does not proceed spontaneously, but requires an
initiating spark or flame. Once begun, the heat produced by the combustion
serves to maintain the reaction until one or both of the reactants are
completely consumed.
Likewise, benzene has a heat of formation of +11.7 kcal/mol, suggesting
that it could decompose spontaneously to hydrogen and carbon. Yet every
chemist knows that benzene is a stable liquid that can be purified by
distillation at 80º C without decomposition. Clearly, many potentially
favorable reactions are prohibited or retarded by substantial energy
barriers to the transformation.
To understand why some reactions occur readily (almost spontaneously),
whereas other reactions are slow, even to the point of being unobservable,
we need to consider the intermediate stages through which reacting
molecules pass on the way to products.
Every reaction in which bonds are broken will necessarily have a higher
energy transition state on the reaction path that must be traversed
before products can form. This is true for both exothermic and endothermic
reactions. In order for the reactants to reach this transition state,
energy must be supplied from the surroundings and reactant molecules must
orient themselves in a suitable fashion. The heat energy needed to raise
the reactants to the transition state energy level is called the
activation enthalpy, ΔH‡. Further treatment of this
subject, and examples of reaction path profiles that illustrate transition
states are provided elsewhere in
this text. However, in these introductory discussions a distinction
between enthalpy and "potential energy" is not made. As expected, the rate
at which chemical reactions proceed is, in large part, inversely
proportional to their activation enthalpies, and is dependent on the
concentrations of the reactants. The study of reaction rates is called
chemical
kinetics.
4. Stability
Common use of the term stability implies an object, system or
situation that is likely to remain unchanged for a significant period of
time. In chemistry, however, we often refer to two kinds of stability.
Thermodynamic Stability : The enthalpy
or potential energy of a compound relative to a reference state. For
exothermic reactions we may say that the products are thermodynamically
more stable than the reactants. The opposite would be true for endothermic
reactions.
Chemical Stability : The resistance of a
compound or mixture of compounds to chemical change (reaction). This is
clearly proportional to the activation energies of all possible reactions.
As noted above, benzene is thermodynamically unstable compared with
elemental carbon and hydrogen, but it is chemically stable under normal
laboratory conditions, even when mixed with some reactive compounds such as
bromine. Compounds or mixtures that are chemically unstable are often
called labile.
When the rates of reactions and equilibria between reactants and products are carefully examined, it becomes apparent that overall enthalpy changes and enthalpy of activation barriers are not by themselves sufficient to explain the observations. A modified energy function, called free energy, is needed. This thermodynamic function is described in the next two sections.
5. Entropy
Enthalpy is not the only thermodynamic function that influences the
overall energy changes, rates and equilibria of chemical and physical
transformations. Two examples will serve to demonstrate this fact.
First, nitrogen pentaoxide is an unstable solid that undergoes a
spontaneous and endothermic decomposition to nitrogen dioxide and
oxygen, as shown below. Clearly, some factor other than a change in
enthalpy must act to favor this decomposition.
|
N2O5 (solid) |
 |
2 NO 2 (gas) + 1/2 O2 (gas) |
ΔHº = +26.2 kcal/mol |
Second, the melting of solids (e.g. ice) and the vaporization of liquids (e.g. water) are endothermic processes. Since intermolecular attractions favor the liquefaction of gases and the solidification of liquids by lowering the enthalpy of the condensed phase, our world would be a frozen lump of solid matter were enthalpy the only controlling factor.
What do these two cases have in common that could
account for their behavior?
In the first example two well-defined molecules in the solid state break
apart into five discrete product molecules, all of them in the gaseous
state. The second example likewise describes a progression from a highly
ordered array of molecules in the solid state to a less ordered assemblage
as a liquid, and finally to a nearly random disordered gaseous state. This
disposition, favoring disorder, seems to be universal, as evidenced by the
failure of a shuffled deck of cards to arrange itself into ordered suits.
The thermodynamic name for this tendency toward randomness or disorder is
entropy, symbol S. Entropy increases spontaneously, since
greater randomness or disorder in a system has a higher statistical
probability. Returning to the deck of cards example, the number of
different ways 52 cards may be arranged is very large (52!), and only a
tiny fraction of these would be ordered in any discernible manner. A
disordered state is therefore more probable than an ordered one.
Since the disorder created by molecular motion increases with temperature,
the units of entropy, eu, are calories per degree Kelvin per mole.
Only differences in entropy have significance, so the standard state
entropy of a substance is the difference between its entropy at 298º K (25º
C) and its entropy at absolute zero (defined as 0 by the third law of
thermodynamics). The standard state entropies of some elements and
compounds are given in the following table. Two obvious trends to note are
that gases have higher entropies than liquids or solids of the same size,
and molecules composed of many atoms have higher entropies than diatomic or
triatomic molecules.
Standard State Entropies, Sº, of Representative Substances
|
Gases |
Liquids |
Solids |
|||
|---|---|---|---|---|---|
| H2 | 31.2 (cal/deg mole) | Br2 | 18.4 (cal/deg mole) | C graphite | 1.4 (cal/deg mole) |
| O2 | 49.0 | H2O | 16.8 | C diamond | 0.6 |
| CO2 | 53.2 | Cyclohexane | 49.2 | I 2 | 14.0 |
| H2O | 45.2 | Benzene | 41.9 | NaCl | 17.2 |
The entropy change in a physical or chemical transformation is written ΔS, and defined as Sproducts - Sreactants. This is the same notation used to represent an enthalpy change, and the sign of ΔS indicates a similar directional change. Thus, a positive ΔS denotes an increase in entropy on going from reactants to products, while a negative sign is associated with a decrease in entropy.
It should be noted that a given sign for ΔSº carries a different
interpretation than it does for ΔHº. Accordingly, a negative ΔHº is
associated with an exothermic and energetically favorable transformation;
but a negative ΔSº indicates an increase in system order (a less random
system), and this is entropically unfavorable. Depending on the case to
which they apply, these two functions may complement (support) each other
or act in opposition. In careful studies of rates and equilibria the
consequence of this relationship must be calculated.
Because entropy increases proportionally with the randomness or statistical
probability of a state, it is useful to identify and describe some common
chemical transformations that result in significant changes in entropy
(|ΔSº| is relatively large). Examples of these will be found at the end of
this chapter.
1. For a given number of total atoms, a process that converts a
few large molecules to many smaller molecules proceeds with a significant
increase in entropy (ΔSº is +).
2.If the total number of molecules remains unchanged, a process
that converts a single molecular species to a mixture of different
molecules proceeds with a significant increase in entropy (ΔSº is +).
3. In general, a change from a condensed phase (solid or liquid)
to a gaseous state will proceed with a significant increase in entropy
(ΔSº is +).
4. Ionization reactions in solution are complicated by solvation
effects. The expected increase in entropy from the first factor
listed above may be offset by solvent molecule orientation about the
ions. This ordering of solvent molecules causes a decrease in ΔSº.
|
The description of entropy given above reflects a traditional or
classic view, which is presently undergoing a significant revision.
This revision focuses on changes in the microscopic distribution of
energy among a large number of translational, rotational and
vibrational quantum states. Instead of the macroscopic concept of
order versus disorder, attention is focused on the concentration or
dispersal of energy in molecular systems. Two fundamental tenets
are that energy spontaneously disperses if not hindered from doing
so (e.g. by activation energy barriers), and that a change in
entropy is a measure of molecular motional energy that has been
dispersed (or concentrated) in a system (as a function of absolute
temperature). |
6. Free Energy
The interaction between enthalpy and entropy changes in chemical reactions is best observed by studying their influence on the equilibrium constants of reversible reactions. To this end a new thermodynamic function called Free Energy (or Gibbs Free Energy), symbol ΔG, is defined as shown in the first equation below. Two things should be apparent from this equation. First, in cases where the entropy change is small, ΔG ≅ ΔH. Second, the importance of ΔS in determining ΔG increases with increasing temperature.
|
ΔGº = ΔHº – TΔSº |
T = temperature in ºK |
The free energy function provides improved insight into the
thermodynamic driving forces that influence reactions. A negative ΔGº is
characteristic of an exergonic reaction, one which is
thermodynamically favorable and often spontaneous, as is the melting of ice
at 1 ºC. Likewise a positive ΔGº is characteristic of an endergonic
reaction, one which requires an input of energy from the
surroundings.
For an example of the relationship of free energy to enthalpy consider the
decomposition of cyclobutane to ethene, shown in the following equation.
The standard state for all the compounds is gaseous.
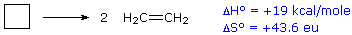
This reaction is endothermic, but the increase in number of molecules
from one (reactants) to two (products) results in a large positive ΔSº.
At 25 ºC (298 ºK), ΔGº = 19 kcal/mol – 298(43.6) cal/mole = 19 – 13
kcal/mole = +6 kcal/mole. Thus, the entropy change opposes the enthalpy
change, but is not sufficient to change the sign of the resulting free
energy change, which is endergonic. Indeed, cyclobutane is perfectly stable
when kept at room temperature.
Because the entropy contribution increases with temperature, this
energetically unfavorable transformation can be made favorable by raising
the temperature. At 200 ºC (473 ºK), ΔGº = 19 kcal/mol – 473(43.6) cal/mole
= 19 – 20.6 kcal/mole = –1.6 kcal/mole. This is now an exergonic
reaction, and the thermal cracking of cyclobutane to ethene is known to
occur at higher temperatures.
|
ΔGº = –RT lnK = –2.303RT logK |
R = 1.987 cal/ ºK mole T = temperature in ºK K = equilibrium constant |
|
A second equation, shown above, is important because it demonstrates the fundamental relationship of ΔGº to the equilibrium constant, K. Because of the negative logarithmic relationship between these variables, a negative ΔGº generates a K>1, whereas a positive ΔGº generates a K<1. When ΔGº = 0, K = 1. Furthermore, small changes in ΔGº produce large changes in K. A change of 1.4 kcal/mole in ΔGº changes K by approximately a factor of 10. This interrelationship may be explored with the calculator on the right. Entering free energies outside the range -8 to 8 kcal/mole or equilibrium constants outside the range 10-6 to 900,000 will trigger an alert, indicating the large imbalance such numbers imply. |
Enter a value in the appropriate box and press "Calculate". |
|||||
7. Free Energy of Activation
Although spontaneous transformations all have negative ΔGº s, not all exergonic processes are spontaneous, due to activation energy barriers to reaction. Our previous treatment of activation energy was framed in terms of enthalpy or potential energy. It should now be clear that, if entropy factors are to be incorporated in the activation barrier, we should be thinking about Free Energy of Activation, ΔG‡ . The defining equation then becomes:
ΔG‡ = ΔH‡ – TΔS‡ |
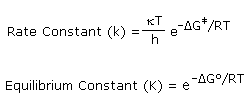
Transition state theory proposes an equilibrium between reactants and
the transition state, so each of the functions in this equation may be
viewed as a [FTransition State –
FReactants] difference, where F represents H, S or G. The
equations on the right demonstrate the similar exponential relationship of
ΔGº to Keq and ΔG‡ to k. Since the rate constant
equation incorporates the temperature variable twice, and ΔG‡
also changes with temperature, observed reaction rates are clearly
temperature dependent. Organic chemists make general use of this
relationship in two ways. First, it is a rule-of-thumb that a 10º C
increase in reaction temperature will roughly double the rate of that
reaction. Second, since this rule applies as well to accompanying
reactions, the rates of such side reactions also increase with temperature,
sometimes more than the desired reaction. Consequently, the practical yield
of the desired product may actually decrease at higher temperatures. Thus,
a cleaner (less contaminated) product is often obtained be running a
reaction at the lowest effective temperature that gives the desired
product.
Because ΔG‡ incorporates a temperature dependent entropy factor
and is related exponentially to the rate constant, k, reaction rate studies
at different temperatures may be interpreted to provide the activation
parameters. Consider, for example, the exergonic substitution reactions
that take place when a 1º-alkyl bromide reacts with sodium thiophenylate,
as shown in the following equation. Careful rate studies made at 20º C in
methanol solution gave the rate constants, k, reported in the second column
of the table below. Enthalpy and entropy of activation values were obtained
by measurements at different temperatures, and are given in the third and
fourth columns of the table.
|
R-CH2-Br + C6H5S(–) Na(+) |
 |
R-CH2-SC6H5 + Na(+) Br(–) |
|
R- |
k |
ΔH‡ |
ΔS‡ |
ΔG‡ kcal/mole |
|---|---|---|---|---|
| H methyl bromide |
100 | +16.2 | -7.5 | +18.4 |
| CH3 ethyl bromide |
39 | +18.1 | -7.7 | +20.35 |
| C2H5 propyl bromide |
25.7 | +17.7 | -10.0 | +20.6 |
| C3H7 butyl bromide |
26.8 | +17.7 | -10.0 | +20.6 |
From this data we can deduce several characteristics of the transition
states through which these simple SN2 reactions must pass. To
begin with, the negative ΔS‡ indicates a transition state that
is more highly ordered than the reactants. This agrees with contemporary
views of the SN2
transition state, in that two independent reactant molecules have
combined to form a single transition complex.
Second, the greater reactivity of methyl bromide over ethyl bromide is
largely due to an increase in ΔH‡, presumably due to steric
hindrance to nucleophile approach from the opposite side of carbon #1.
Finally, the decrease in reaction rates of 1-propyl and 1-butyl bromides
compared with ethyl bromide, while less pronounced and nearly the same in
each case, is due primarily to an increasingly negative ΔS‡ . It
has been proposed that this comes from a restriction of conformational
mobility about the C2-C3 bond of the alkyl group in the transition state.
Of three possible staggered conformations, on average one will interfere
significantly with nucleophile approach, resulting in a one third lower
probability for achieving the transition complex.
In this respect it should be noted that entropy has been related to the
statistical probability of a system, W (the number of equivalent way
molecules can be arranged) by the expression: S = κ*lnW (κ is the Boltzman
constant = R/Avogadro's Number)
Some applications of the concepts and principles presented here to specific chemical systems are presented in the topics accessed from the following menu.