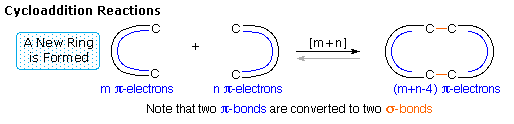Pericyclic Reactions
Pericyclic Reactions
An important body of chemical reactions, differing from ionic or free
radical reactions in a number of respects, has been recognized and
extensively studied. Among the characteristics shared by these reactions,
three in particular set them apart.:
1. They are relatively unaffected by solvent changes, the presence
of radical initiators or scavenging reagents, or (with some exceptions) by
electrophilic or nucleophilic catalysts.
2. They proceed by a simultaneous (concerted) series of bond
breaking and bond making events in a single kinetic step, often with high
stereospecificity.
3. In agreement with 1 & 2, no ionic, free radical
or other discernible intermediates lie on the reaction path.
Since reactions of this kind often proceed by nearly simultaneous reorganization of bonding electron pairs by way of cyclic transition states, they have been termed pericyclic reactions. The four principle classes of pericyclic reactions are termed: Cycloaddition, Electrocyclic, Sigmatropic, and Ene Reactions. A general illustration of each class will be displayed by clicking on the following diagram. The cycloaddition and ene reactions are shown in their intermolecular format. Corresponding intramolecular reactions, which create an additional ring, are well known.
All these reactions are potentially reversible (note the gray arrows).
The reverse of a cycloaddition is called cycloreversion and proceeds
by a ring cleavage and conversion of two sigma-bonds to two pi-bonds. The
electrocyclic reaction shown above is a ring forming process. The reverse
electerocyclic ring opening reaction proceeds by converting a sigma-bond to
a pi-bond. As shown, the retro ene reaction cleaves an unsaturated compound
into two unsaturated fragments. Finally, sigmatropic bond shifts may
involve a simple migrating group, as shown in the example above, or may
take place between two pi-electron systems (e.g. the Cope
rearrangement).
The general descriptions shown above provide a basis for reaction
classification, but care must be taken to assure that a given
transformation is truly concerted. Unfortunately, this is not a trivial
determination, often requiring a combination of isotope labeling and
stereochemical studies to arrive at a plausible conclusion. There is also a
subtle distinction to be made between a synchronous reaction in
which all bond-making and bond-breaking events take place in unison, and a
multi-stage concerted process in which some events precede others
without generating an intermediate state.
Although some pericyclic reactions occur spontaneously, most require the introduction of energy in the form of heat or light, with a remarkable product dependence on the source of energy used. An appreciation of the stereoselective structural changes these reactions promote is best achieved by inspecting some individual examples.
1. Cycloaddition Reactions
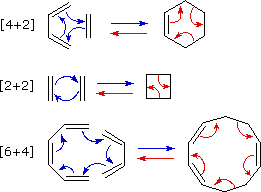 A concerted
combination of two π-electron systems to form a ring of atoms having two
new σ bonds and two fewer π bonds is called a cycloaddition reaction. The
number of participating π-electrons in each component is given in brackets
preceding the name, and the reorganization of electrons may be depicted by
a cycle of curved
arrows - each representing the movement of a pair of electrons. These
notations are illustrated in the drawing on the right. The ring-forming
cycloaddition reaction is described by blue arrows, whereas the
ring-opening cycloreversion process is designated by red arrows.
Note that the number of curved arrows needed to show the bond
reorganization is half the number total in the brackets.
A concerted
combination of two π-electron systems to form a ring of atoms having two
new σ bonds and two fewer π bonds is called a cycloaddition reaction. The
number of participating π-electrons in each component is given in brackets
preceding the name, and the reorganization of electrons may be depicted by
a cycle of curved
arrows - each representing the movement of a pair of electrons. These
notations are illustrated in the drawing on the right. The ring-forming
cycloaddition reaction is described by blue arrows, whereas the
ring-opening cycloreversion process is designated by red arrows.
Note that the number of curved arrows needed to show the bond
reorganization is half the number total in the brackets.
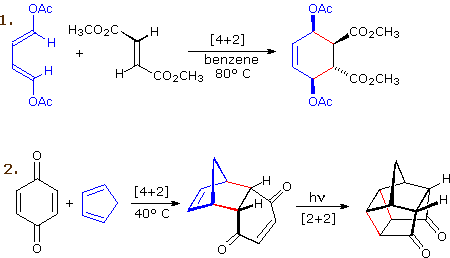
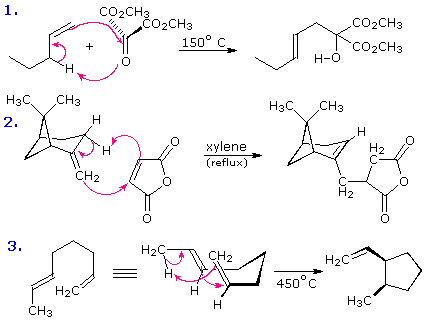
The most common cycloaddition reaction is the [4π+2π]
cyclization known as the Diels-Alder
reaction. In Diels-Alder terminology the two reactants are referred to
as the diene and the dienophile. The following diagram shows
two examples of [4π+2π] cycloaddition, and in the
second equation a subsequent light induced [2π+2π]
cycloaddition. In each case the diene reactant is colored blue, and the new
σ-bonds in the adduct are colored red. The stereospecificity of these
reactions should be evident. In the first example, the acetoxy substituents
on the diene have identical E-configurations, and they remain cis to
each other in the cyclic adduct. Likewise, the ester substituents on the
dienophile have a trans-configuration which is maintained in the adduct.
The reactants in the second equation are both monocyclic, so the
cycloaddition adduct has three rings. The orientation of the quinone
six-membered ring with respect to the bicycloheptane system (colored blue)
is endo, which
means it is oriented cis to the longest or more unsaturated bridge. The
alternative configuration is called exo.
Since the dienophile (quinone) has two activated double bonds, a second
cycloaddition reaction is possible, provided sufficient diene is supplied.
The second cycloaddition is slower than the first, so the monoadduct shown
here is easily prepared in good yield. Although this [4+2] product is
stable to further heating, it undergoes a [2+2] cycloaddition when exposed
to sunlight. Note the loss of two carbon-carbon π-bonds and the formation
of two σ-bonds (colored red) in this transformation. Also note that the
pi-subscript is often omitted from the [m+n] notation for the majority of
cycloadditions involving π-electron systems.
By clicking on this diagram two more examples of cycloaddition reactions will be displayed. Reaction 3 is an intramolecular Diels-Alder reaction. Since the diene and dienophile are joined by a chain of atoms, the resulting [4+2] cycloaddition actually forms two new rings, one from the cycloaddition and the other from the linking chain. Once again the addition is stereospecific, ignoring the isopropyl substituent, the ring fusion being cis and endo. The fourth reaction is a [6+4] cycloaddition.
2. Electrocyclic Reactions
 An electrocyclic
reaction is the concerted cyclization of a conjugated π-electron system by
converting one π-bond to a ring forming σ-bond. The reverse reaction may be
called electrocyclic ring opening. Two examples are shown on the right. The
electrocyclic ring closure is is designated by blue arrows, and the ring
opening by red arrows. Once again, the number of curved arrows that
describe the bond reorganization is half the total number of electrons
involved in the process.
An electrocyclic
reaction is the concerted cyclization of a conjugated π-electron system by
converting one π-bond to a ring forming σ-bond. The reverse reaction may be
called electrocyclic ring opening. Two examples are shown on the right. The
electrocyclic ring closure is is designated by blue arrows, and the ring
opening by red arrows. Once again, the number of curved arrows that
describe the bond reorganization is half the total number of electrons
involved in the process.
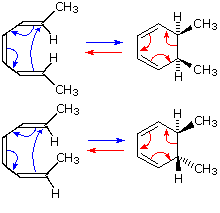
In the first case, trans,cis,trans-2,4,6-octatriene undergoes
thermal ring closure to cis-5,6-dimethyl-1,3-cyclohexadiene. The
sterospecificity of this reaction is demonstrated by closure of the
isomeric trans,cis,cis-triene to
trans-5,6-dimethyl-1,3-cyclohexadiene, as noted in the second
example.
By clicking on this diagram two examples of
thermal electrocyclic opening of cyclobutenes to conjugated butadienes will
be displayed. This mode of reaction is favored by relief of ring strain,
and the reverse ring closure (light blue arrows) is not normally observed.
Photochemical ring closure can be effected, but the stereospecificity is
opposite to that of thermal ring opening.
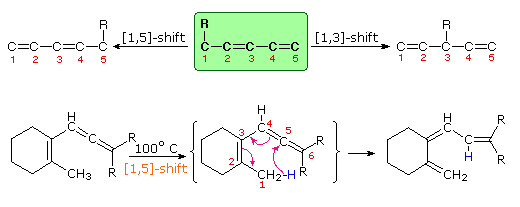
3. Sigmatropic Rearrangements
Molecular rearrangements in which a σ-bonded atom or group, flanked by one or more π-electron systems, shifts to a new location with a corresponding reorganization of the π-bonds are called sigmatropic reactions. The total number of σ-bonds and π-bonds remain unchanged. These rearrangements are described by two numbers set in brackets, which refer to the relative distance (in atoms) each end of the σ-bond has moved, as illustrated by the first equation in the diagram below. The most common atom to undergo sigmatropic shifts is hydrogen or one of its isotopes. The second equation in the diagram shows a facile [1,5] hydrogen shift which converts a relatively unstable allene system into a conjugated triene. Note that this rearrangement, which involves the relocation of three pairs of bonding electrons, may be described by three curved arrows.
By clicking on this diagram two additional
examples of thermal [1,5] hydrogen shifts will be displayed. These
reactions are particularly informative in that [1,3] hydrogen shifts are
not observed. The reactant in the first equation is a deuterium labeled
1,3,5-cyclooctatriene. On heating, this compound equilibrates with its
1,3,6-triene isomer, and the two deuterium atoms are scrambled among the
four locations noted. If [1,3] or [1,7] hydrogen shifts were taking place,
the deuterium atoms would be distributed equally among all eight carbon
atoms. On prolonged heating, or at higher temperatures these
cyclooctatrienes undergo electrocyclic ring opening to 1,3,5,7-octatetraene
and reclosure to vinyl-1,3-cyclohexadienes.
The second example shows another [1,5] hydrogen shift, from the proximal
methyl group to the carbonyl oxygen atom. The resulting dienol rapidly
exchanges OH for OD before the [1,5] shift reverses. In this manner the
reactive methyl is soon converted to CD3. Since hydrogens alpha
to a carbonyl group are known to undergo acid or base
catalyzed exchange by way of enol intermediates, we might expect the
α'-CH2 group to exchange as well. However, if care is taken to
remove potential acid or base catalysts, the thermal [1,3] shift necessary
for the exchange is found to be very slow.
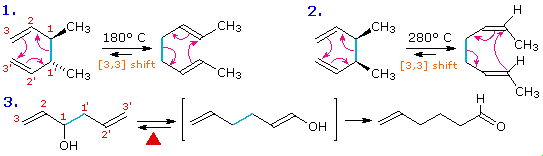
The [3,3] sigmatropic rearrangement of 1,5-dienes or allyl vinyl ethers,
known respectively as the Cope and Claisen rearrangements, are among
the most commonly used sigmatropic reactions. Three examples of the Cope
rearrangement are shown in the following diagram. Reactions 1 and
2 (top row) demonstrate the stereospecificity of this reaction. The
light blue σ-bond joins two allyl groups, oriented so their ends are near
each other. Since each allyl segment is the locus of a [1,3] shift, the
overall reaction is classified as a [3,3] rearrangement. The three
pink colored curved arrows describe the redistribution of three bonding
electron pairs in the course of this reversible rearrangement. The diene
reactant in the third reaction is drawn in an extended conformation. This
molecule must assume a coiled conformation (as above) before the [3,3]
rearrangement can take place. The product of this rearrangement is an enol
which immediately tautomerizes to its keto form. Such variants are termed
the oxy-Cope rearrangement, and are useful because the reverse
rearrangement is blocked by rapid ketonization. If the hydroxyl substituent
is converted to an alkoxide salt, the activation energy of the
rearrangement is lowered significantly.
The degenerate or self-replicating Cope rearrangement has been a
fascinating subject of research. For examples
.
Two examples of the Claisen Rearrangement may be seen by clicking on the above diagram. Reaction 4. is the
classic rearrangement of an allyl phenyl ether to an ortho-allyl phenol.
The methyl substituent on the allyl moiety serves to demonstrate the
bonding shift at that site. The initial cyclohexadienone product
immediately tautomerizes to a phenol, regaining the stability of the
aromatic ring. Reaction 5 is an aliphatic analog in which a vinyl
group replaces the aromatic ring. In both cases three pairs of bonding
electrons undergo a reorganization.
By clicking on the above diagram a second time
two examples of [2,3] sigmatropic rearrangements will be displayed. The
allylic sulfoxide in reaction 6 rearranges reversibly to a less
stable sulfenate ester. The weak S-O bond may be reductively cleaved by
trimethyl phosphite to an allylic alcohol and a thiol (not shown). Reaction
7 shows a similar rearrangement of a sulfur
ylide to a cyclic sulfide. The [2,3]-Wittig
rearrangement is yet another example.
4. Ene Reactions
The joining of a double or triple bond to an alkene reactant having a transferable allylic hydrogen is called an ene reaction. The reverse process is called a retro ene reaction. In the bonding direction the ene reaction is characterized by the redistribution of three pairs of bonding electrons. and may be described by a cycle of three curved arrows. As noted earlier, this bond reorganization involves the overall conversion of a π-bond to a σ-bond (or the opposite in the case of retro ene fragmentation). This is the same bond bookkeeping change exhibited by electrocyclic reactions, but no rings are formed or broken in an ene reaction unless it is intramolecular. The following examples illustrate some typical ene reactions, with equation 3 being an intramolecular ene reaction. Ene reactions are favored when the hydrogen accepting reagent, the "enophile", is electrophilic. This is the case for reactions 1 and 2, which proceed under milder conditions than 3, despite the latter's intramolecular nature.
Hydrogen is the most common atom transferred in an ene reaction. Indeed,
all the examples shown above involve hydrogen shifts. Other atoms or groups
may, however, participate in ene-like transformations. Two such cases will
be displayed above by clicking on the diagram.
Reaction 4 is drawn as a retro ene reaction, although this has not been
demonstrated to be general for all reactions of allylic alcohols with
thionyl chloride. Equation 5 illustrates an unusual "magnesium ene
reaction" in which a Grignard function moves to a new location before
reacting with an electrophilic reagent such as CO2. Because this
is an intramolecular ene reaction a new ring is formed. Clicking on the diagram a second time will display two
additional examples. Equation 6 demonstrates that an enol tautomer, even in
low concentration, may function as the hydrogen donor in the ene reaction.
Equation 7 is one of many examples of Lewis acid catalysis in the ene
reaction. A similar acid-catalyzed reaction of simple aldehydes with
alkenes to give allylic alcohols, 1,3-diols or 1,3-dioxanes is known as the
Prins reaction.
Certain retro ene reactions have proven useful as synthetic
transformations. Examples of these concerted elimination reactions may be
examined by Clicking
Here
A more elaborate treatment of the intramolecular ene reaction is available
by Clicking
Here
5. Stereochemical Notations
One characteristic shared by most pericyclic reactions, and noted in
many cases described above, is their stereospecificity. This is not the
first class of reactions for which a characteristic stereospecificity has
been noted. Substitution
reactions may proceed randomly or by "inversion" or "retention" of
configuration. Elimination
reactions may occur in an "anti" or "syn" fashion, or may be
configurationally random. The terms "syn" and "anti" have also been applied
to 1,2-addition
reactions.
Since these configurational change notations are not appropriate for
pericyclic reactions, new designations are needed. Cycloaddition
reactions and sigmatropic rearrangements both involve pairs of
σ-bond-making events (or a coupled bond-making & bond-breaking)
associated with a π-electron system. If all the bonding events take place
on the same face of the π-system the configuration of the reaction is
termed suprafacial. If the bonding events occur on opposite sides or
faces of the π-system the reaction is termed antarafacial.
Suprafacial examples of these pericyclic transformations are shown below.
The bracketed numbers that designate reactions of this kind sometimes carry
subscripts (s or a) that specify their configuration. Thus the
cycloaddition on the left may be termed a [4s + 2s]
process.
Although cycloaddition reactions are concerted (no intermediate species are
formed), the two new bonds are not necessarily formed in a synchronous
fashion. Depending on partial charge distribution in the diene and
dienophile reactants, the formation of one bond may lead the development of
the other. Such unsymmetrical transition state bonding is termed
asynchronous.
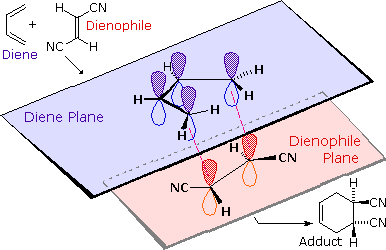 |
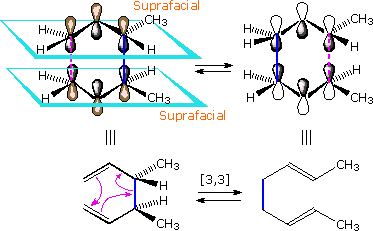 |
|
| A Suprafacial [4+2] Cycloaddition | A Suprafacial [3,3] Sigmatropic Rearrangement |
|---|
To see a model of a Diels-Alder transition state .
An example of an antarafacial [1,7] hydrogen shift is shown in the following diagram. The conjugated triene assumes a nearly planar coiled conformation in which a methyl hydrogen is oriented just above the end carbon atom of the last double bond. A [1s,7a] sigmatropic hydrogen shift may then take place, as described by the four curved arrows. With reference to the approximate plane of this π-electron system (defined by the green bonds), the hydrogen atom departs from the bottom face and bonds to the top face, so the transfer is antarafacial.
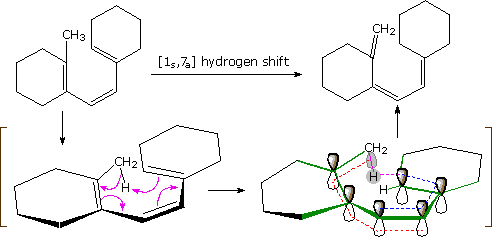
A different notation for configurational change is required for electrocyclic reactions. In these cases a σ-bond between the ends of a conjugated π-electron system is either made or broken with a corresponding loss or gain of a π-bond. For this to happen, the terminal carbon atoms of the conjugated π-electron system must be rehybridized with an accompanying rotation or twisting of roughly 90º. When viewed along the axis of rotation, the two end groups may turn in the same direction, termed conrotatory, or in opposite directions, termed disrotatory. The prefixes con and dis may be remembered by association with their presence in the words concur & disagree. These two modes of electrocyclic reaction are shown in the following diagram in the general form in which they are most commonly observed. Specific examples of these electrocyclic reactions were given earlier.
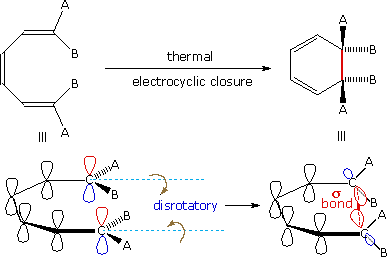 |
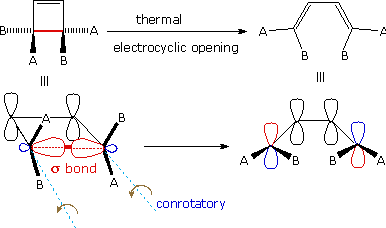 |
|
| A Disrotatory Electrocyclic Closure | A Conrotatory Electrocyclic Opening |
|---|
To see an animation of conrotatory electrocyclic ring closure .
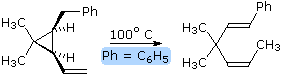 Since ene
reactions usually involve coupled bond-making & bond-breaking
operations associated with short π-electron systems (2 or 3 carbons), their
stereospecificity is almost always suprafacial with respect to both
components. This configurational feature is illustrated by the retro ene
equation on the right. By clicking on the
diagram, a representation of the transition state for this
stereospecific transformation will be drawn. Note that bond-breaking and
bond-making takes place in a suprafacial orientation with respect to each
π-electron system. This reaction is facilitated by the relief of small ring
strain.
Since ene
reactions usually involve coupled bond-making & bond-breaking
operations associated with short π-electron systems (2 or 3 carbons), their
stereospecificity is almost always suprafacial with respect to both
components. This configurational feature is illustrated by the retro ene
equation on the right. By clicking on the
diagram, a representation of the transition state for this
stereospecific transformation will be drawn. Note that bond-breaking and
bond-making takes place in a suprafacial orientation with respect to each
π-electron system. This reaction is facilitated by the relief of small ring
strain.
Several different structural relationships between the reacting moieties of an intramolecular ene reaction are possible. The examples shown here and above represent the most common orientation. To see examples of two other arrangements Click Here.
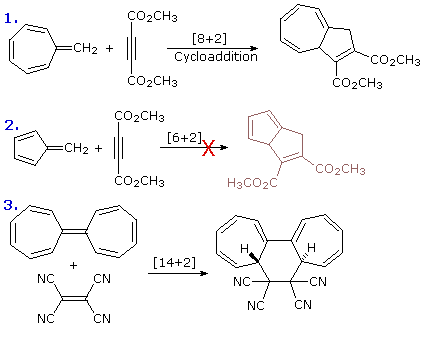
6. Perplexing Features of Pericyclic Reactions
The examples of pericyclic reactions presented here provide ample
evidence of their usefulness in constructing or modifying complex
molecules, often with a high degree of stereospecificity. However, in
contrast to the general applicability of most common ionic reactions,
pericyclic reactions often display a marked sensitivity to small structural
changes. Thus, stereospecificity may flip-flop, and rates may vary a
million fold or more. In the case of cycloaddition reactions, the three
equations on the right illustrate this fact. Equations 1 and
2 show two very similar transformations, but the first takes place
with moderate heat and the second does not. Note that in each case the
triple bond only contributes two electrons to the cycloaddition transition
state. The common [4+2] cycloaddition known as the Diels-Alder reaction
proceeds stereospecifically in a suprafacial fashion,
but the [14+2] cycloaddition in equation 3 is antarafacial with
respect to the polyene.
Electrocyclic and sigmatropic reactions also show puzzling differences in
behavior. By clicking on the diagram four
examples will be shown. Equations 4 and 5 describe similar
electrocyclic ring openings of stereoisomeric cyclobutenes. The first
occurs under relatively mild heating, but the second requires extreme heat
and may well proceed by bond homolysis to a diradical. Equation 6
shows two electrocyclic ring closures of
trans,cis,trans-2,4,6-octatriene. The thermal reaction is
disrotatory, and the photochemical process is conrotatory.
Finally, the absence of [1,3] sigmatropic shifts of hydrogen was noted earlier, and a clear example is shown in equation
7. Isomerization of the conjugated triene to toluene should be
strongly exothermic, but a concerted rearrangement of this kind would be a
[1,3] sigmatropic process. In the absence of acid catalysts this triene is
completely stable to moderate heating. Any [1,5] hydrogen shifts that take
place reform the starting triene and would require isotopic labeling to
prove. Of course, the isomerization to toluene occurs rapidly if acid is
added.
7. A Useful Mnemonic Rule
Before pericyclic reactions can be put to use in a predictable and controlled manner, a broad mechanistic understanding of the factors that influence these concerted transformations must be formulated. The simplest, albeit least rigorous, method for predicting the configurational path favored by a proposed pericyclic reaction is based upon a transition state electron count. In most of the earlier examples, pericyclic reactions were described by a cycle of curved arrows, each representing a pair of bonding electrons. The total number of electrons undergoing reorganization is always even, and is either a 4n+2 or 4n number (where n is an integer). Once this electron count is made, the following table may be used for predictions.
|
Thermal |
Transition State Class |
Configurational Preference |
|---|---|---|
| 4n + 2 (aromatic) | Suprafacial or Disrotatory | |
| 4n (antiaromatic) | Antarafacial or Conrotatory | |
|
Photochemical |
Transition State Class |
Configurational Preference |
| 4n + 2 (aromatic) | Antarafacial or Conrotatory | |
| 4n (antiaromatic) | Suprafacial or Disrotatory |
Although this modest mnemonic does not make explicit use of molecular orbitals, more rigorous methods that are founded on the characteristics of such orbitals have provided important insight into these reactions. Since pericyclic reactions proceed by a cyclic reorganization of bonding electron pairs, it is necessary to evaluate changes in the associated molecular orbitals that take place in going from reactants to products. The following section describes approaches of this kind.
Theoretical Models for Pericyclic Reactions
Theoretical Models for Pericyclic Reactions
In 1965 R. B. Woodward and Roald Hoffmann of Harvard University proposed and demonstrated that concerted reactions proceed most readily when there is congruence between the orbital symmetries of the reactants and products. In other words, when the bonding character of all occupied molecular orbitals is preserved at all stages of a concerted molecular reorganization, that reaction will most likely take place. The greater the degree of bonding found in the transition state for the reaction, the lower will be its activation energy and the greater will be the reaction rate.
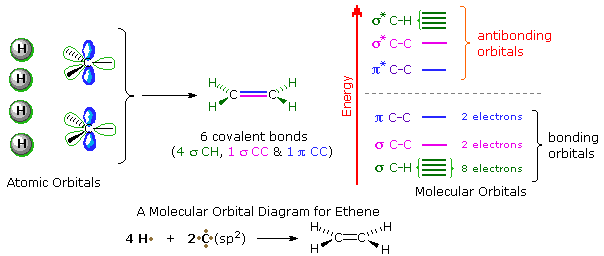
A general introduction to molecular orbitals was presented earlier. The simple compound ethene is made up of six atoms held together by six covalent bonds, as described in the following illustration. A molecular orbital diagram of ethene is created by combining the twelve atomic orbitals associated with four hydrogen atoms and two sp2 hybridized carbons to give twelve molecular orbitals. Six of these molecular orbitals (five sigma & one pi-orbital) are bonding, and are occupied by the twelve available valence shell electrons. The remaining six molecular orbitals are antibonding, and are empty.
Proper molecular orbitals are influenced by all the nuclei in a molecule, and require consideration of the full structure and symmetry of a molecule for their complete description. For most purposes, this level of treatment is not needed, and more localized orbitals serve well. In the case of ethene and other isolated double bonds, descriptions of the localized π orbitals will be displayed by clicking on the above diagram. Several important characteristics of molecular orbitals need to be pointed out, and this diagram will serve to illustrate them.
1. The spatial distribution of electron density for most
occupied molecular orbitals is discontinuous, with regions of high
density separated by regions of zero density, e.g. a nodal plane.
The π-orbital on the left has one nodal plane (colored light blue), and
the π*-orbital on the right has a second nodal plane (colored
yellow). As a rule, higher energy molecular orbitals have a larger number
of nodal surfaces or nodes.
2. The wave functions that describe molecular orbitals undergo a
change in sign at nodal surfaces. This phase change is sometimes
designated by plus and minus signs associated with discrete regions of
the orbital, but this notation may sometimes be confused for an electric
charge. In the above diagram, regions having one phase sign are colored
blue, while those having an opposite sign are colored red.
3. These localized orbitals may be classified by two independent
symmetry
operations ; a mirror plane perpendicular to the functional plane and
bisecting the the molecule (colored yellow above), and a two-fold axis of
rotation (C2) created by the intersection of this mirror plane
with the common nodal plane (colored light blue). The π-orbital on the
left is symmetric (S) with respect to the mirror plane, but
antisymmetric (A) when rotated 180º, a C2 operation.
The opposite is true for the π*-orbital on the right, which
has a mirror plane symmetry of A and a C2 symmetry of
S. Such symmetry characteristics play an important role in
creating the orbital diagrams used by Woodward and Hoffmann to
rationalize pericyclic reactions.
A model of the p and π orbitals of a double bond may be examined by .
The original approach of Woodward and Hoffmann involved construction of an "orbital correlation diagram" for each type of pericyclic reaction. The symmetries of the appropriate reactant and product orbitals were matched to determine whether the transformation could proceed without a symmetry imposed conversion of bonding reactant orbitals to antibonding product orbitals. If the correlation diagram indicated that the reaction could occur without encountering such a symmetry-imposed barrier, it was termed symmetry allowed. If a symmetry barrier was present, the reaction was designated symmetry-forbidden. Two related methods of analyzing pericyclic reactions are the transition state aromaticity approach, and the frontier molecular orbital approach. Each of these methods has merit, and a more detailed description of each may be examined by clicking the appropriate button below.
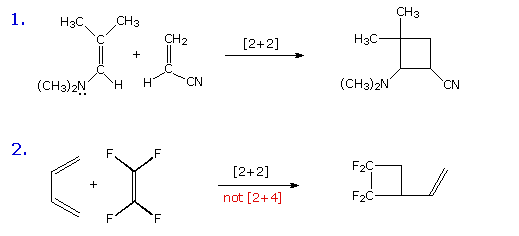
1. Some Examples
Before reviewing representative examples of various types of pericyclic reactions, the previous caution that a given transformation be truly concerted must be emphasized again. The two equations shown in the following diagram describe [2+2] cycloaddition reactions. The second example is particularly interesting because a [4+2] Diels-Alder cycloaddition is possible, but provides only a minor product. A careful examination of these reactions, using probes for ionic and radical intermediates, has shown that these are not concerted transformations. The dipolar and diradical intermediates proposed for these reactions will be illustrated by clicking on the diagram.
By clicking on the above diagram a second time, an apparent electrocyclic ring opening reaction will be shown. The symmetry favored conrotatory concerted path would generate a very strained trans-cyclohexene double bond, and is energetically unlikely. Instead, a higher activation energy bond cleavage to a diradical intermediate takes place on heating. The racemic diastereomer of this compound undergoes the same ring opening at a lower temperature, and this is believed to be a concerted conrotatory electrocyclic reaction..
With this caveat in mind, extensive lists of pericyclic reactions may be
assembled, and their rationalization by the previously noted mnemonic or
orbital analysis is both remarkably successful and instructive. Many of the
reactions cited earlier, together with additional examples, will be
displayed by clicking on the appropriate button.
| Click Here. |
|---|
More about Pericyclic ReactionsAn outstanding treatment of pericyclic reactions, including
lots of challenging questions with answers, has been provided by
Henry Rzepa. |