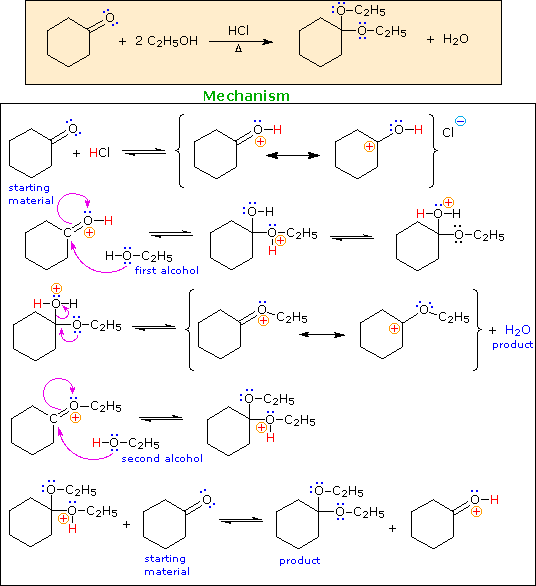
The Mechanism of Acetal Formation
Acetal derivatives of aldehydes and ketones are prepared by an
acid-catalyzed dehydration reaction with alcohols or diols. An example is
shown below.
Writing a mechanism for this reaction provides a good test of ones'
understanding of acid-catalyzed processes.
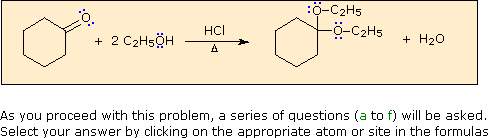
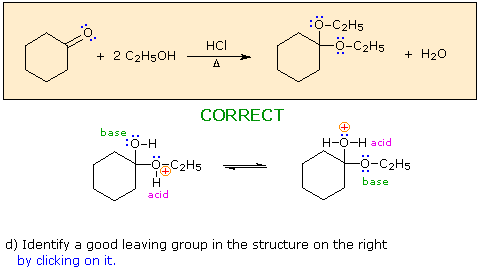
a) The first step in this process
must be an acid-base proton transfer.
Click on the basic atom that is protonated to initiate
this transformation.
When you have made a correct selection, an equation showing the reaction for that step will appear, and a new question will be posed. Incorrect selections will open an alert window displaying an explanatory message. The overall transformation will remain displayed in the colored box at each stage.
The Mechanism of Acetal Formation
The Mechanism of Acetal Formation
The Mechanism of Acetal Formation
The Mechanism of Acetal Formation
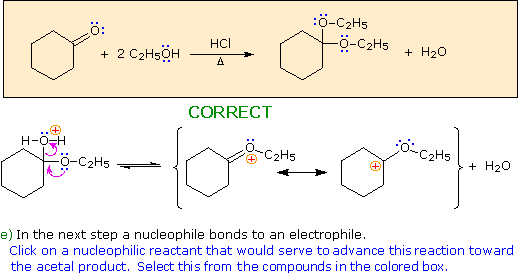
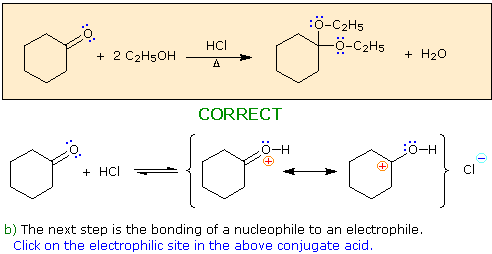
Choose a nucleophile from the reactants in the colored box.
The Mechanism of Acetal Formation
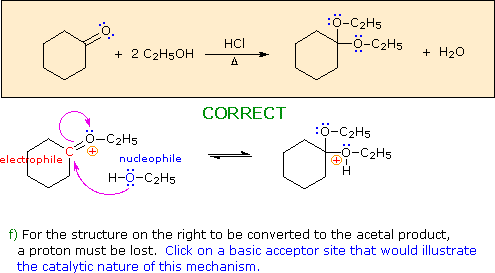
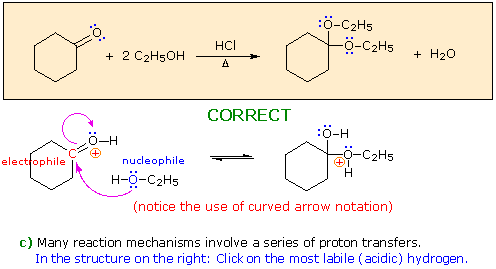
Hint: Choose a base from the reactants in the colored box.
Acetal Formation
Congratulations. You have finished this problem. A summary of the steps in this acid-catalyzed mechanism is shown below.