The following questions are organized somewhat by functional group (as
many organic courses introduce nomenclature one functional group at a
time). The index below will take you to the beginning of questions that
focus on a particular functional group, or you may page through the
document as you like. If you are asked to write a IUPAC name for a compound, pay careful
attention to the syntax of this nomenclature system. For example, commas
and dashes must be used in precisely the correct manner, and extra spaces
must be avoided. Spelling must also be absolutely correct. Computers (and
some instructors) are unforgiving when it comes to judging the names you
submit. Part A: Enter the number of carbons indicated by each of the
following root names in the designated answer box. Part B: The line formula for a branched alkane is shown
below. Part C: The line formula for another branched alkane is shown
below. Part D: The formula for a substituted cycloalkane is shown
below. Part E: The formula for another cycloalkane derivative is shown
below. Part A: The line formula for a branched alkene is shown
below. Part B: The line formula for a cyclic alkene is shown below. Part C: Name the following compounds according to the IUPAC
system: Part A:
(CH3)2CHCH(C2H5)CH2CH2CH3 Part B:
CH2=C(C2H5)CH2CH2CH3 Part C:
(CH3)2C=CHC(C2H5)=CH2 Part D: CH3C≡CCH(C2H5)2 Part E: (CH3)3CC≡CCH2CH=C(CH3)2 Name the following compounds according to the IUPAC system: Name the following compounds according to the IUPAC system:Organic Nomenclature Problems
Question 1:Basic Hydrocarbon
NomenclatureQuestion 2:Unsaturated
Hydrocarbon NomenclatureQuestion 3:Nomenclature from Condensed FormulasQuestion
4:Nomenclature of Aromatic
HydrocarbonsQuestion 5:Nomenclature of
Halogen CompoundsQuestion 6:Nomenclature of
AlcoholsQuestion 7:Nomenclature of Aldehydes
& KetonesQuestion 8:Nomenclature of
Carboxylic Acids
Question 1: Basic Hydrocarbon
Nomenclature
Root
Number of Carbons
Root
Number of Carbons
Hexane
Propane
Butane
Pentane
Nonane
Heptane
Decane
Ethane
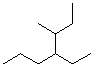
What is the molecular formula of this
compound?
CH
How many carbon atoms are in the longest
chain?
How many alkyl substituents are attached to this chain?
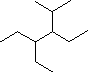
In the IUPAC system what is the root or base name
of this compound?
How many alkyl substituents are attached to the
longest chain?
Give the IUPAC name for this compound.
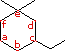
In the IUPAC system what is the root or base name
of this compound?
How many alkyl substituents are attached to the
ring?
In numbering the ring, which carbon (a-f) is #1?
In numbering the ring, which carbon (a-f) is #2?
Give the IUPAC name for this compound.

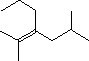
Give the IUPAC name for this compound.
Question 2: Unsaturated Hydrocarbon
Nomenclature
What is the molecular formula of this
compound?
CH
How many carbon atoms are in the longest chain,
ignoring the double bond?
What is the longest chain incorporating both
carbons of the double bond?
How many substituents are on this chain?
Give the IUPAC name for this compound.
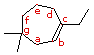
In numbering the ring, which carbon (a-g) is #1?
Give the IUPAC name for this compound.
(1)

Enter the IUPAC name
(2)
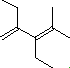
Enter the IUPAC name
(3)

Enter the IUPAC name
(4)

Enter the IUPAC name
(5)
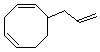
Enter the IUPAC name
(6)
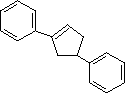
Enter the IUPAC name
Question 3: Hydrocarbon Nomenclature from
Condensed Formulas
Give the IUPAC name for this compound.
Give the IUPAC name for this compound.
Give the IUPAC name for this compound.
Give the IUPAC name for this compound.
Give the IUPAC name for this compound.
Question 4: Aromatic Hydrocarbons
(1)
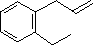
Enter the IUPAC name
(2)
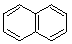
Enter the IUPAC name
(3)
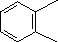
Enter the IUPAC name
(4)
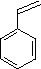
Enter the IUPAC name
(5)
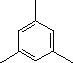
Enter the IUPAC name
Question 5: Halogen Substituted
Compounds
(1)
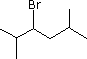
Enter the IUPAC name
(2)
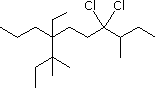
Enter the IUPAC name
(3)
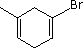
Enter the IUPAC name
(4)
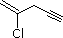
Enter the IUPAC name
(5)
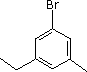
Enter the IUPAC name
whreusch@pilot.msu.edu.