Spectroscopy
Introduction to Spectroscopy
In previous sections of this text the structural formulas of hundreds of organic compounds have been reported, often with very little supporting evidence. These structures, and millions of others described in the scientific literature, are in fact based upon sound experimental evidence, which was omitted at the time in order to focus on other aspects of the subject. Much of the most compelling evidence for structure comes from spectroscopic experiments, as will be demonstrated in the following topics.
The Light of Knowledge is an often used phrase, but it is particularly appropriate in reference to spectroscopy. Most of what we know about the structure of atoms and molecules comes from studying their interaction with light (electromagnetic radiation). Different regions of the electromagnetic spectrum provide different kinds of information as a result of such interactions. Realizing that light may be considered to have both wave-like and particle-like characteristics, it is useful to consider that a given frequency or wavelength of light is associated with a "light quanta" of energy we now call a photon. As noted in the following equations, frequency and energy change proportionally, but wavelength has an inverse relationship to these quantities.
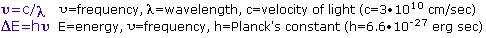
In order to "see" a molecule, we must use light having a wavelength
smaller than the molecule itself (roughly 1 to 15 angstrom units). Such
radiation is found in the X-ray region of the spectrum, and the field of
X-ray crystallography yields remarkably detailed pictures of
molecular structures amenable to examination. The chief limiting factor
here is the need for high quality crystals of the compound being studied.
The methods of X-ray crystallography are too complex to be described here;
nevertheless, as automatic instrumentation and data handling techniques
improve, it will undoubtedly prove to be the procedure of choice for
structure determination.
The spectroscopic techniques described below do not provide a
three-dimensional picture of a molecule, but instead yield information
about certain characteristic features. A brief summary of this information
follows:
• Mass Spectrometry: Sample molecules are ionized by high
energy electrons. The mass to charge ratio of these ions is measured very
accurately by electrostatic acceleration and magnetic field perturbation,
providing a precise molecular weight. Ion
fragmentation patterns may be related to the structure of the molecular
ion.
• Ultraviolet-Visible Spectroscopy: Absorption of this relatively
high-energy light causes electronic excitation. The easily accessible
part of this region (wavelengths of 200 to 800 nm) shows absorption only
if conjugated pi-electron systems are
present.
• Infrared Spectroscopy: Absorption of this lower energy radiation
causes vibrational and rotational excitation of groups of atoms. within
the molecule. Because of their characteristic absorptions identification of functional groups is easily
accomplished.
• Nuclear Magnetic Resonance Spectroscopy: Absorption in the
low-energy radio-frequency part of the spectrum causes excitation of
nuclear spin states. NMR spectrometers are tuned to certain nuclei (e.g.
1H, 13C, 19F & 31P). For
a given type of nucleus, high-resolution
spectroscopy distinguishes and counts atoms in different locations in the
molecule.
Table of Contents
|
Mass Spectrometry |
||||
| The Mass Spectrometer | ||||
| Characteristics of Mass Spectra | ||||
| Isotopes | ||||
| Fragmentation Patterns | ||||
| High Resolution Spectra | ||||
|
Ultraviolet-Visible Spectroscopy |
||||
| Background | ||||
| The Electromagnetic Spectrum | ||||
| UV-Visible Absorption Spectra | ||||
| The Importance of Conjugation | ||||
|
Infrared Spectroscopy |
||||
| Introduction | ||||
| Vibrational Spectroscopy | ||||
| Group Frequencies | ||||
|
Nuclear Magnetic Resonance Spectroscopy (NMR) |
||||
| Introduction | ||||
| Proton NMR Spectroscopy | ||||
| The Chemical Shift | ||||
| Signal Strength | ||||
| Spin-Spin Splitting | ||||
| Carbon NMR Spectroscopy | ||||
|
At various points throughout this text, links to supplementary information or special topics will be located in shaded boxes of this kind. |
|---|
Spectroscopy Resources
| Mass Spectrometry Tutorial | Prepared at the University of Arizona. Many instructive problems are provided. | |
| Modern Methods of Mass Spectrometry | An excellent introduction to modern methods of mass spectrometry from University of Leeds. | |
| NMR Tutorial and Spectroscopy Problems | Prepared at Imperial College. A more advanced tutorial than that provided in this text. The associated problems all involve nmr spectroscopy, but also use data from infrared and mass spectrometry. | |
| NMR Spectroscopy-A Virtual Text | Prepared by Joseph Hornak, Rochester Institute of Technology. An exceptional introduction to modern nmr spectroscopy. More advanced than this text. | |
| NMR meets Musicians | A clever and informative site from the University of Erlangen-Nuremberg, Institute of Organic Chemistry | |
| Spectroscopy Tools | A useful site providing information for NMR, IR and mass spectroscopy. Created at the University of Potsdam. | |
| More Spectroscopy Tools | A nice collection of applications for interpreting NMR, IR and mass spectra. Created at Colby College. | |
| Spectra Data Base | A large and very useful collection of MS, NMR, IR, Raman & ESR spectra. This page is hosted by SDBS, Japan. | |
Spectroscopy Problems |
||
|---|---|---|
| 64 Graded Problems | Compiled at Notre Dame. A very nice collection of problems using all the spectroscopy methods discussed here. | |
| 72 Graded Problems | Compiled at UCLA. An outstanding collection of unknowns requiring the interpretation of nmr spectra & some ir. Solutions are provided. A general discussion of nmr and its uses is given. Some advanced techniques not discussed in this text are used. | |
| Infrared Spectroscopy Problems | Prepared at Colby College. This is the Netscape version. | |